Users
Protocol Co-ordinator or Investigator who is in charge of patient screening at testing facility.
Objective
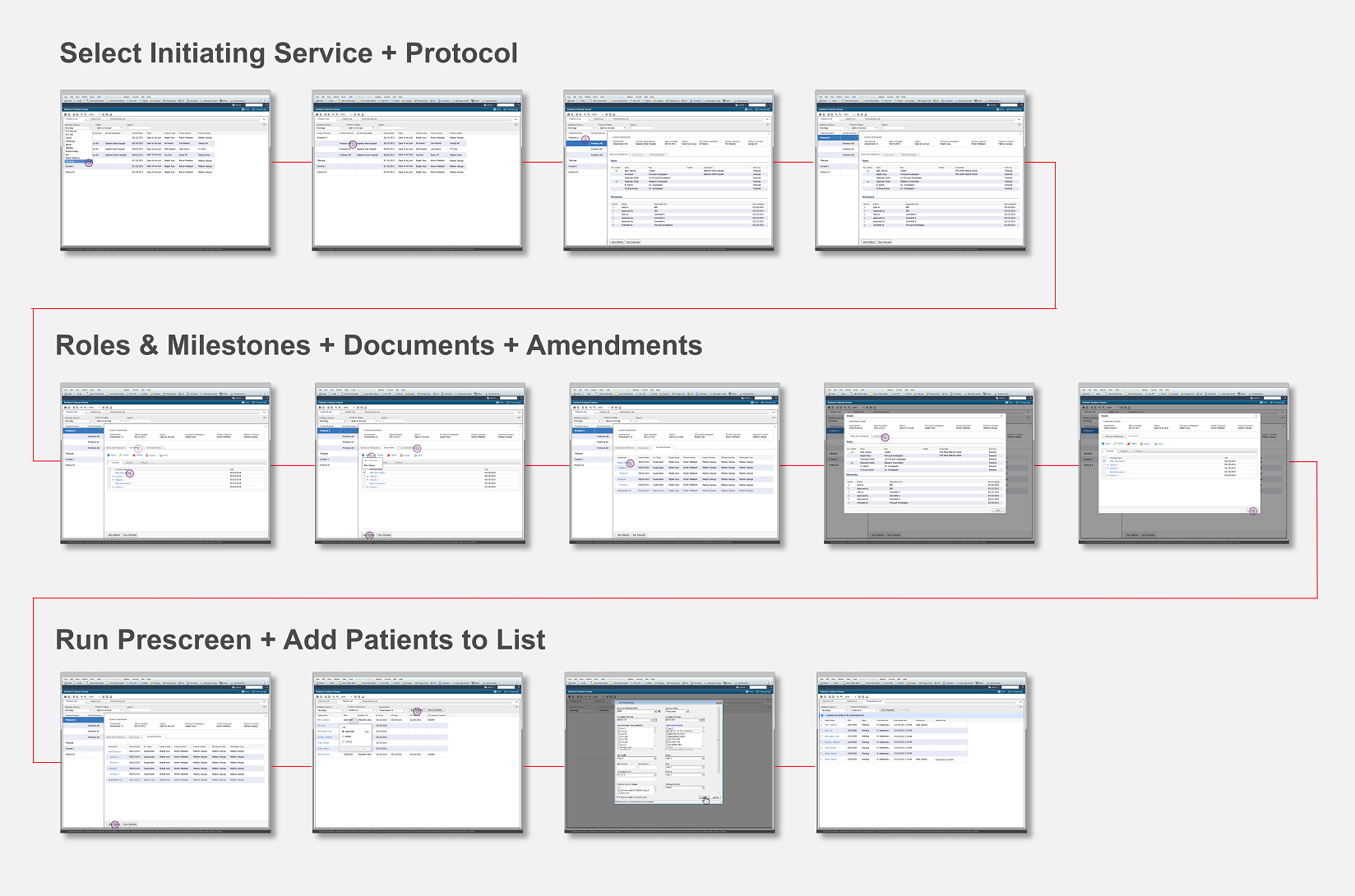
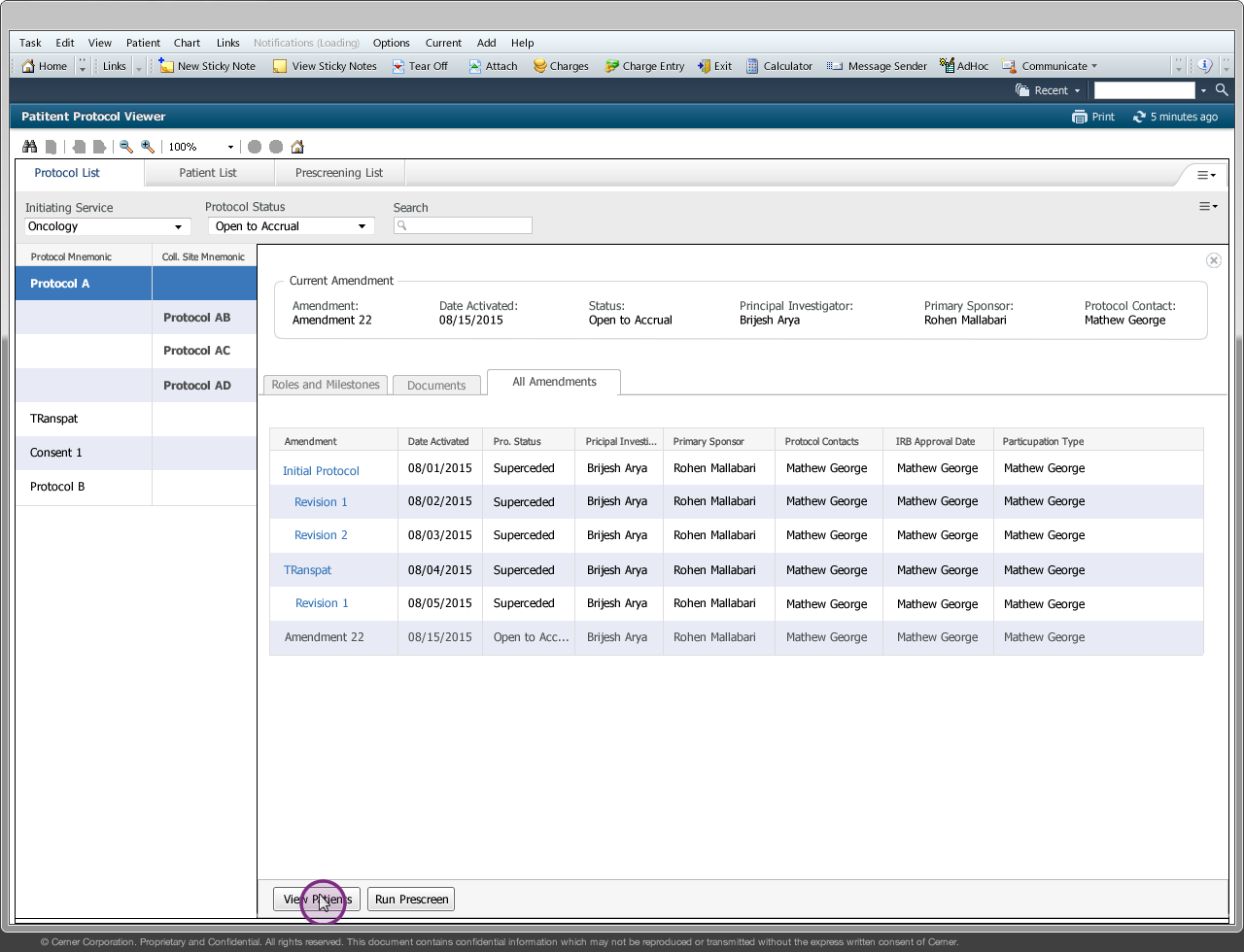
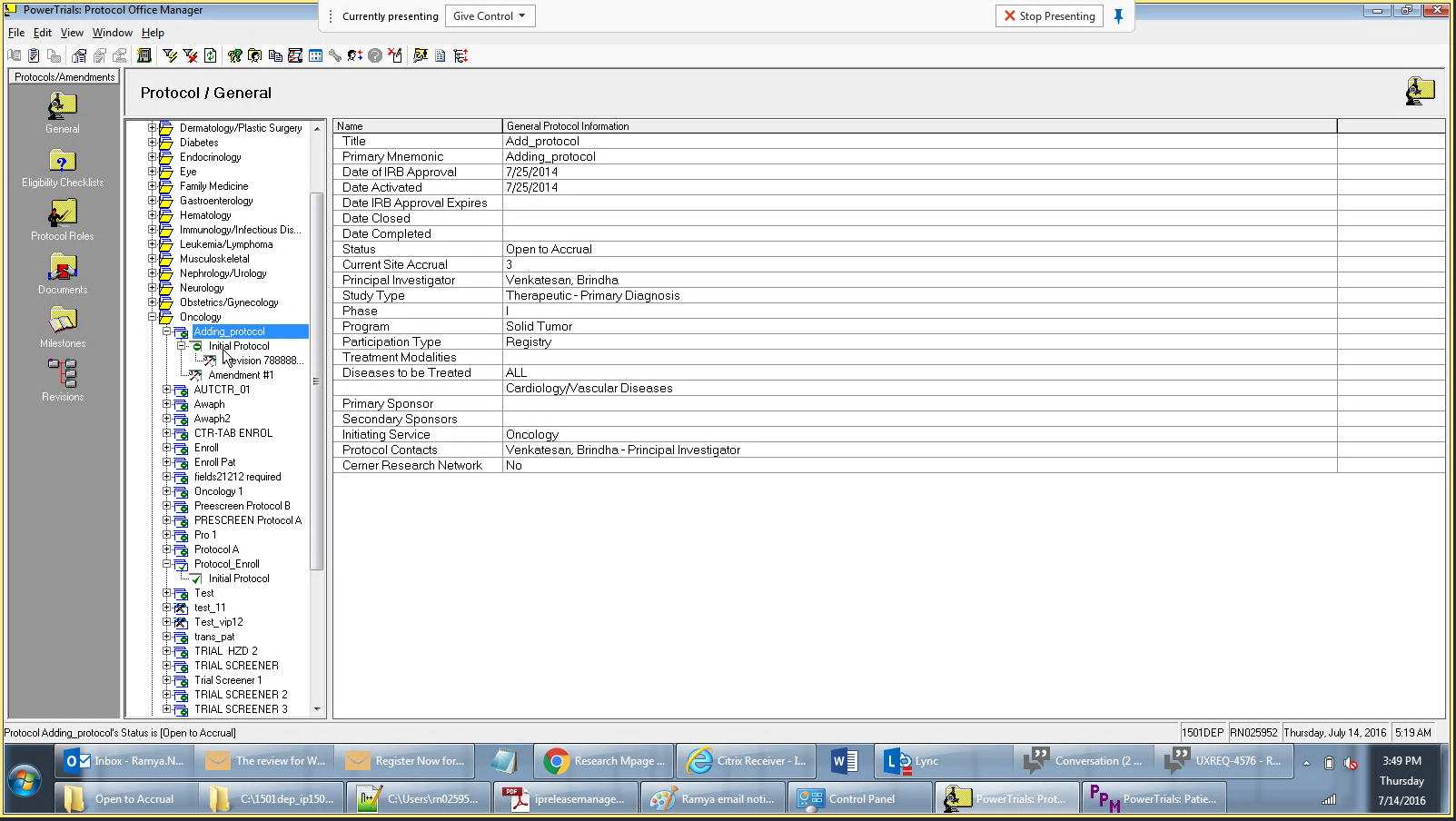
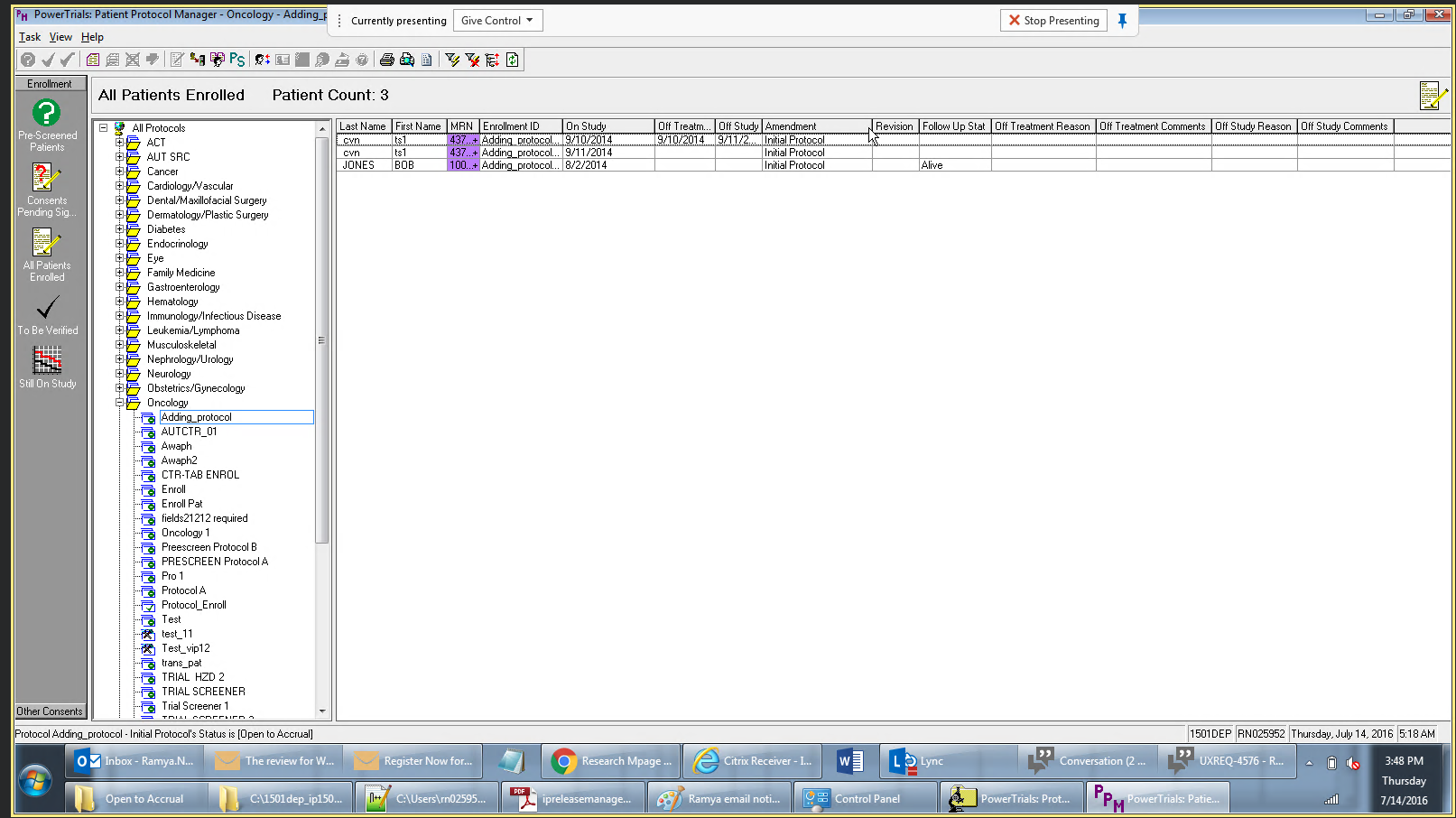
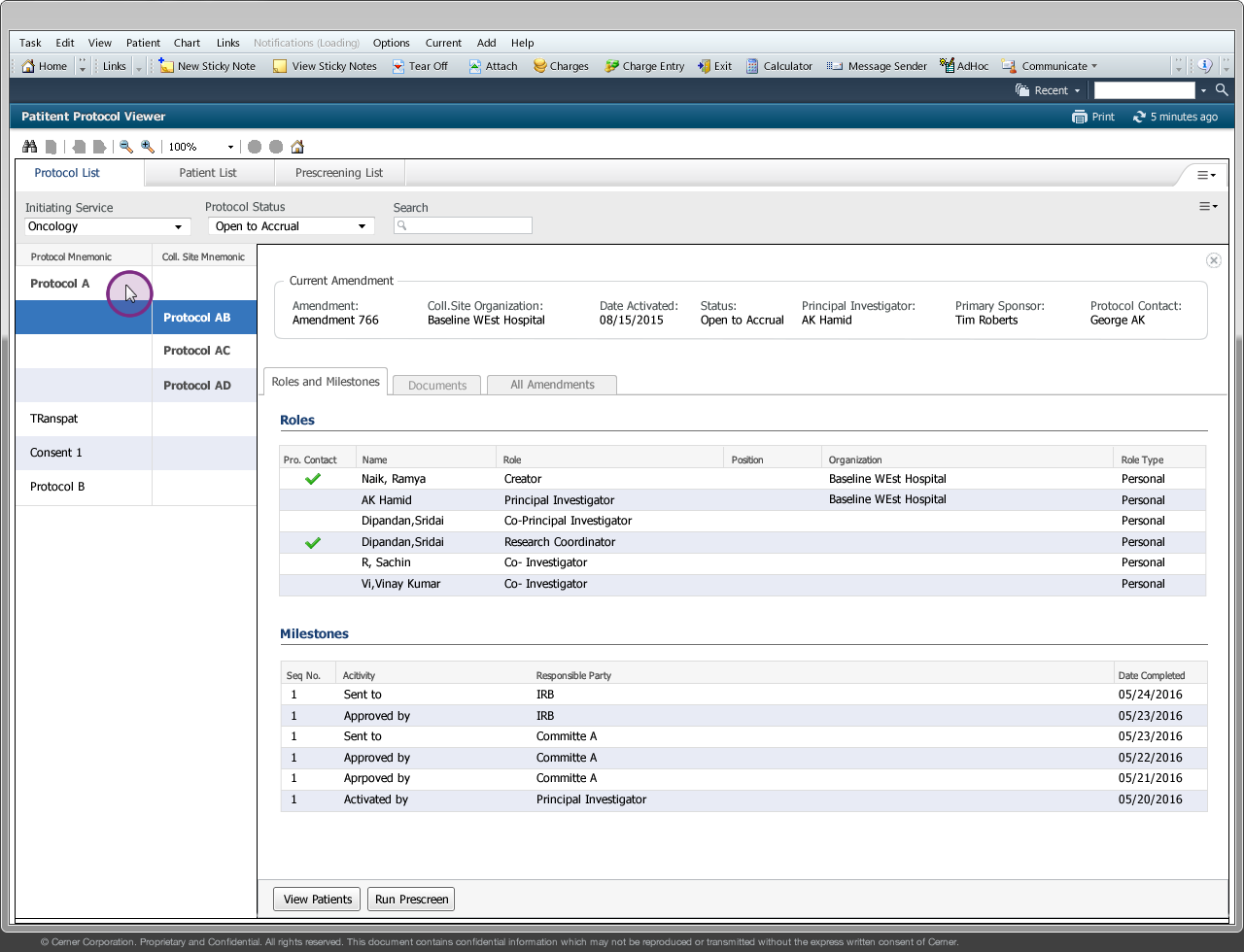
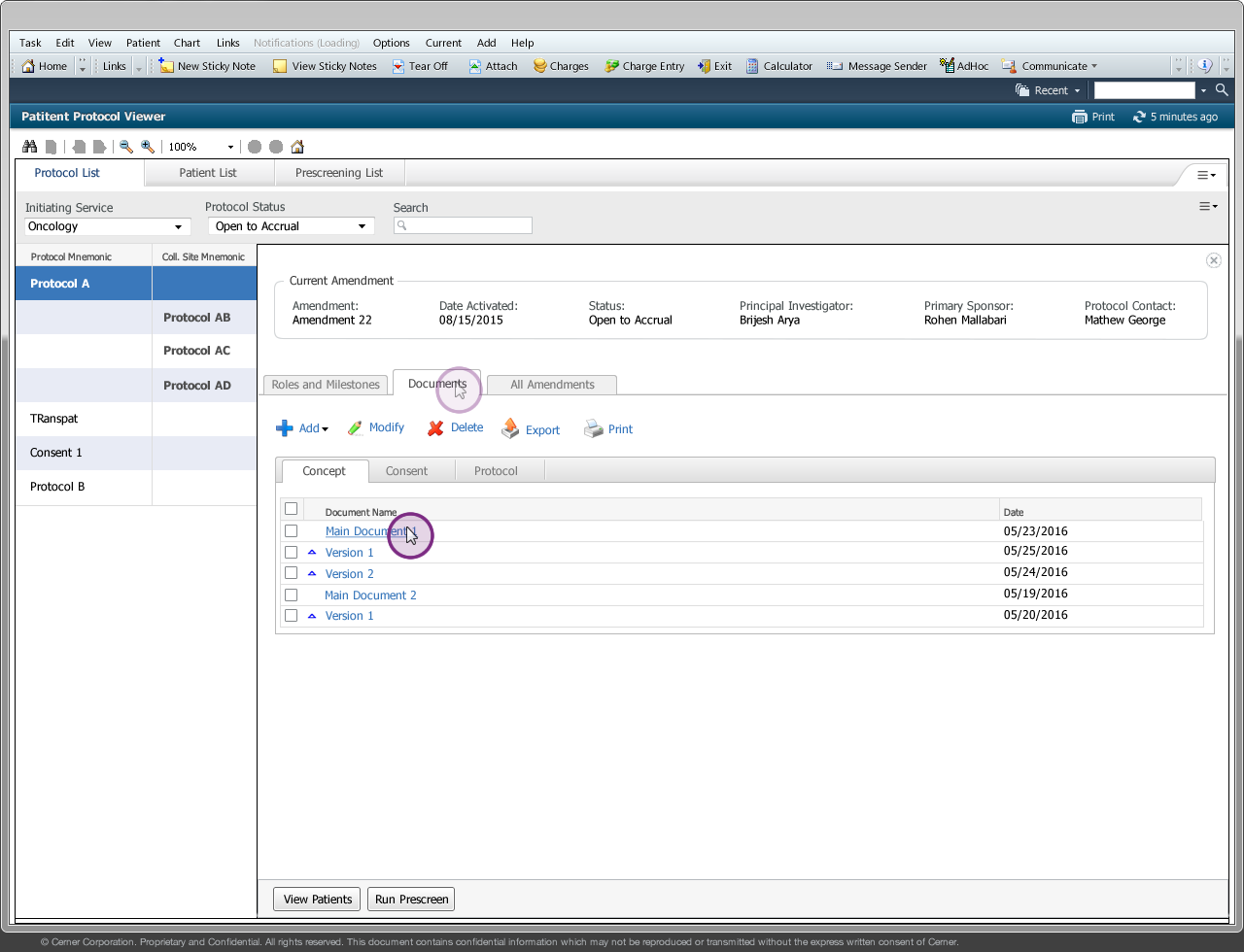
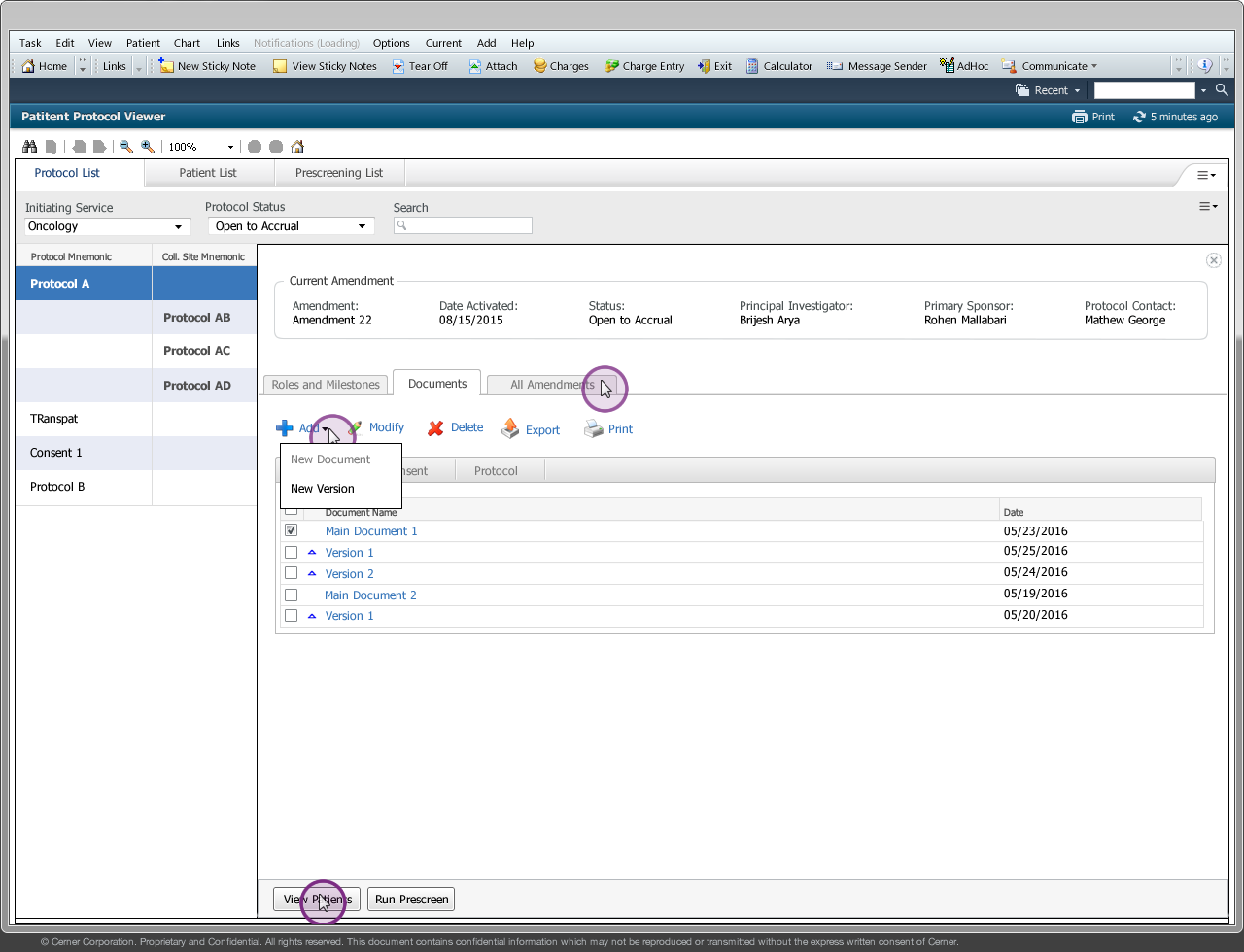
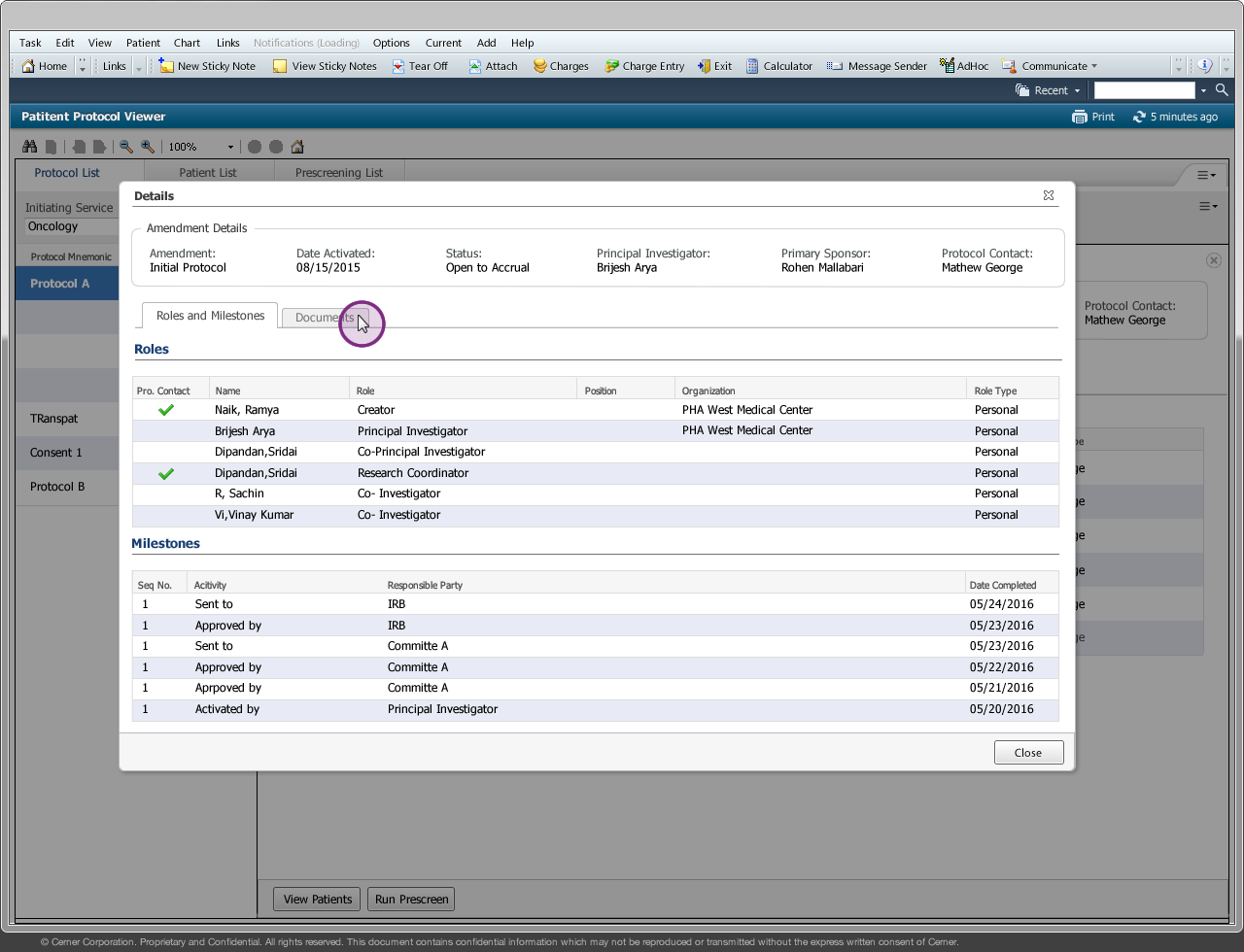
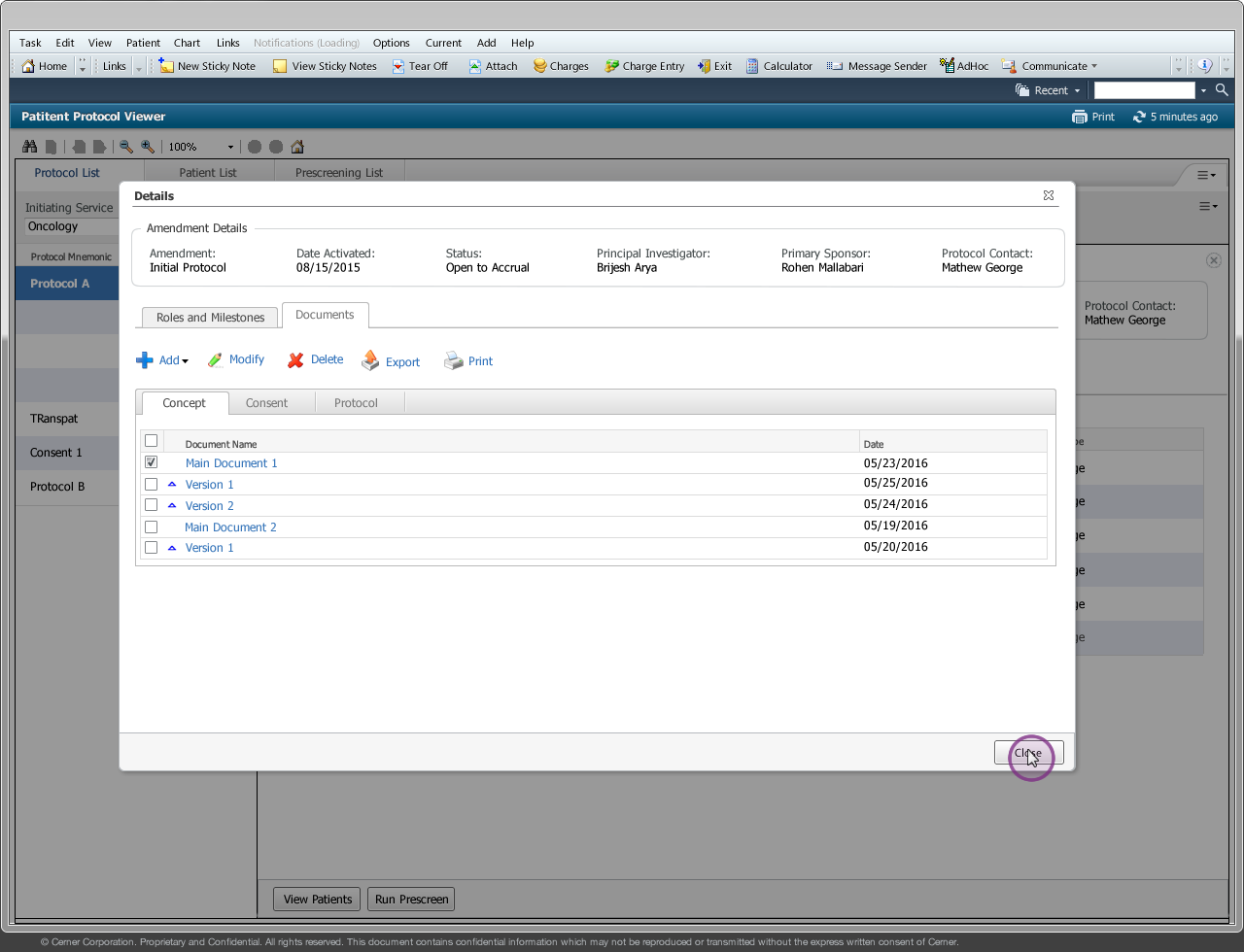
Drug companies manufacture drugs that should be tested for effects on patients before mass production. Patient Protocol Manager application has a list of protocols with eligible patient list based on specific criteria, who then undergo a clinical drug test. This application can also host documents like patient consent forms, protocol forms, protocol roles and milestones and previous amendments from individual patients and run eligibility criteria tests that can append new patients to the protocol list.
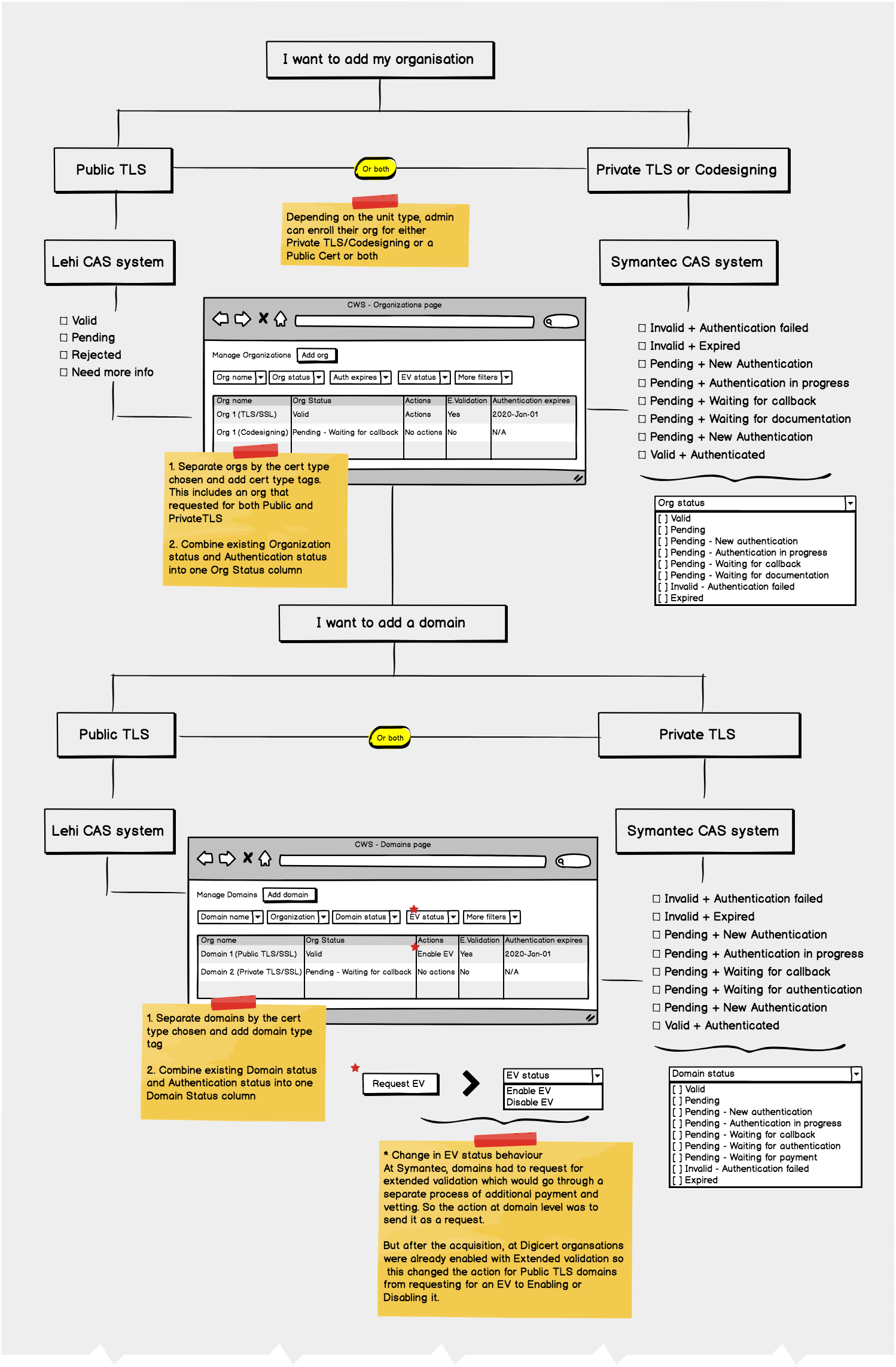
Protocol - Protocol is an instance of a new drug test
Amendment/Revision - Changes made to a protocol will be considered as an amendment or revision. The decision to consider it as an amendment or revision is within the Clinical Trial Board. There are no limits to the number of amendments or revision a protocol can have but only one amendment/revision can be in ‘Open to Accrual’ state.
Technology Platform
Finding the right platform for the new application was a tedious task. Considerable amount of time was spent to understand commonly used platforms and find the right fit based on application’s requirements. Multiple meetings with the platform and application stakeholders ensured that we were on the right track with the selection.
My Role
Visual and Interaction Designer, collaborating with Product owners on solution understanding while working with engineering and PM team for implementation strategy
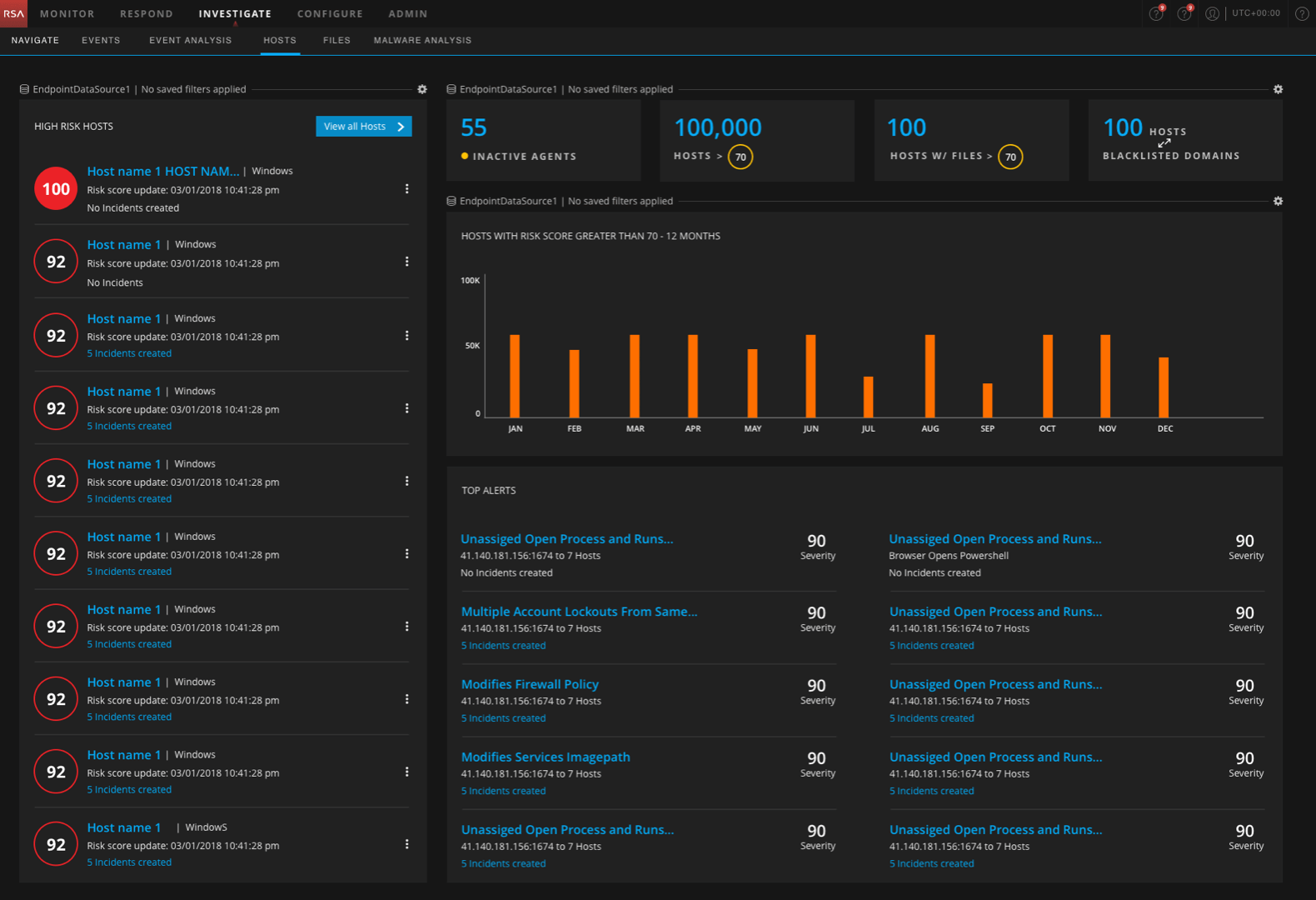
PowerTrials Protocol Manager
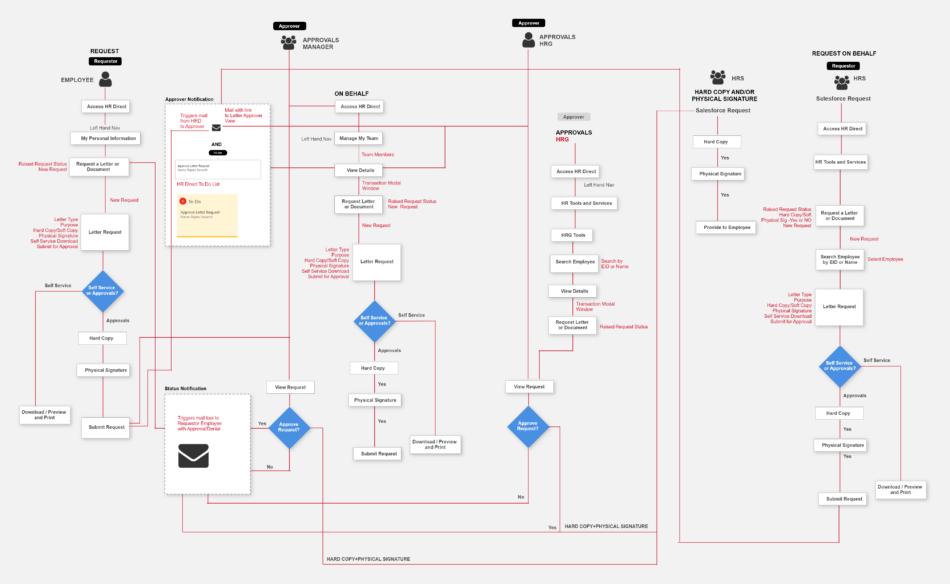
Task Flow
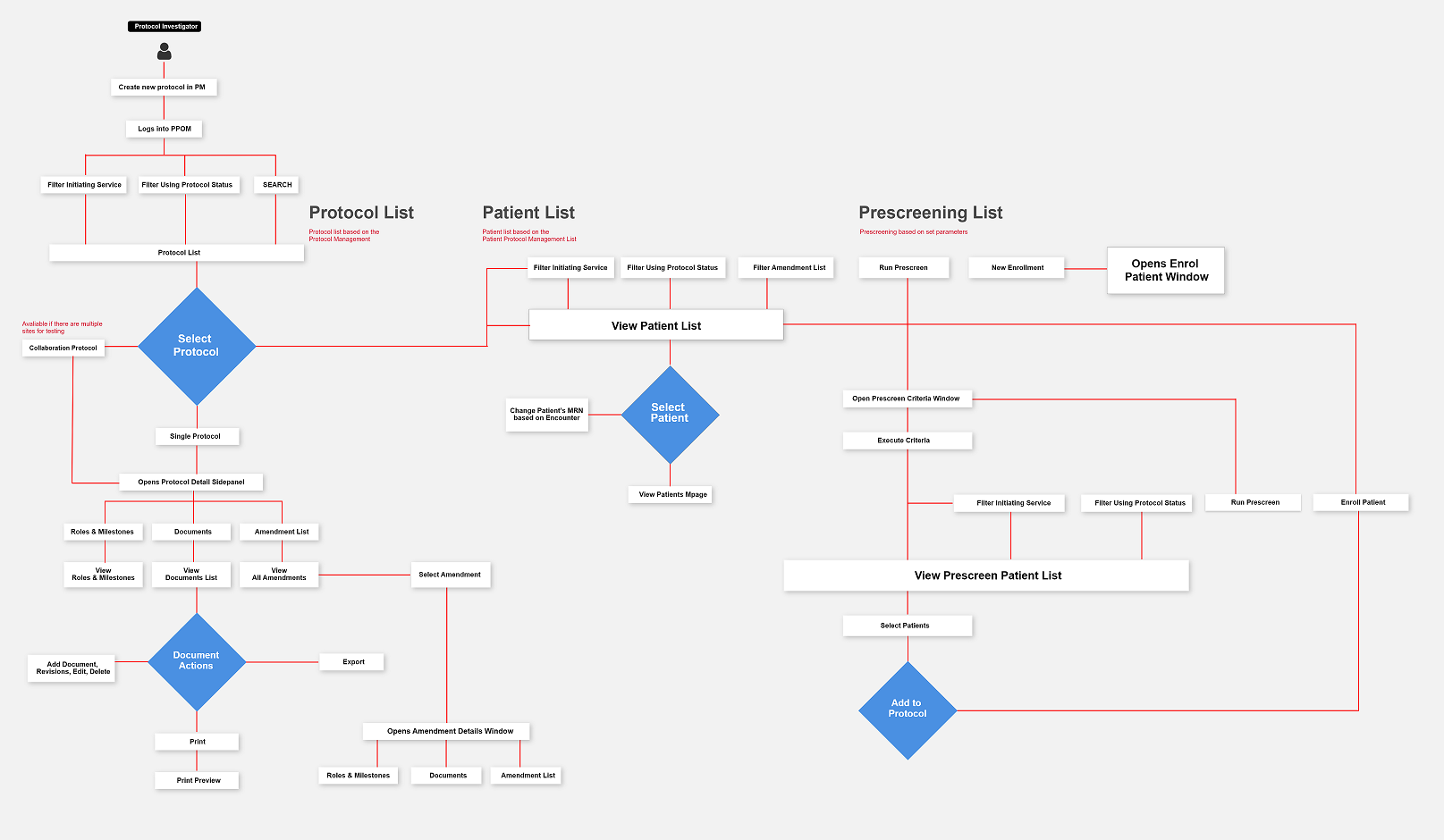
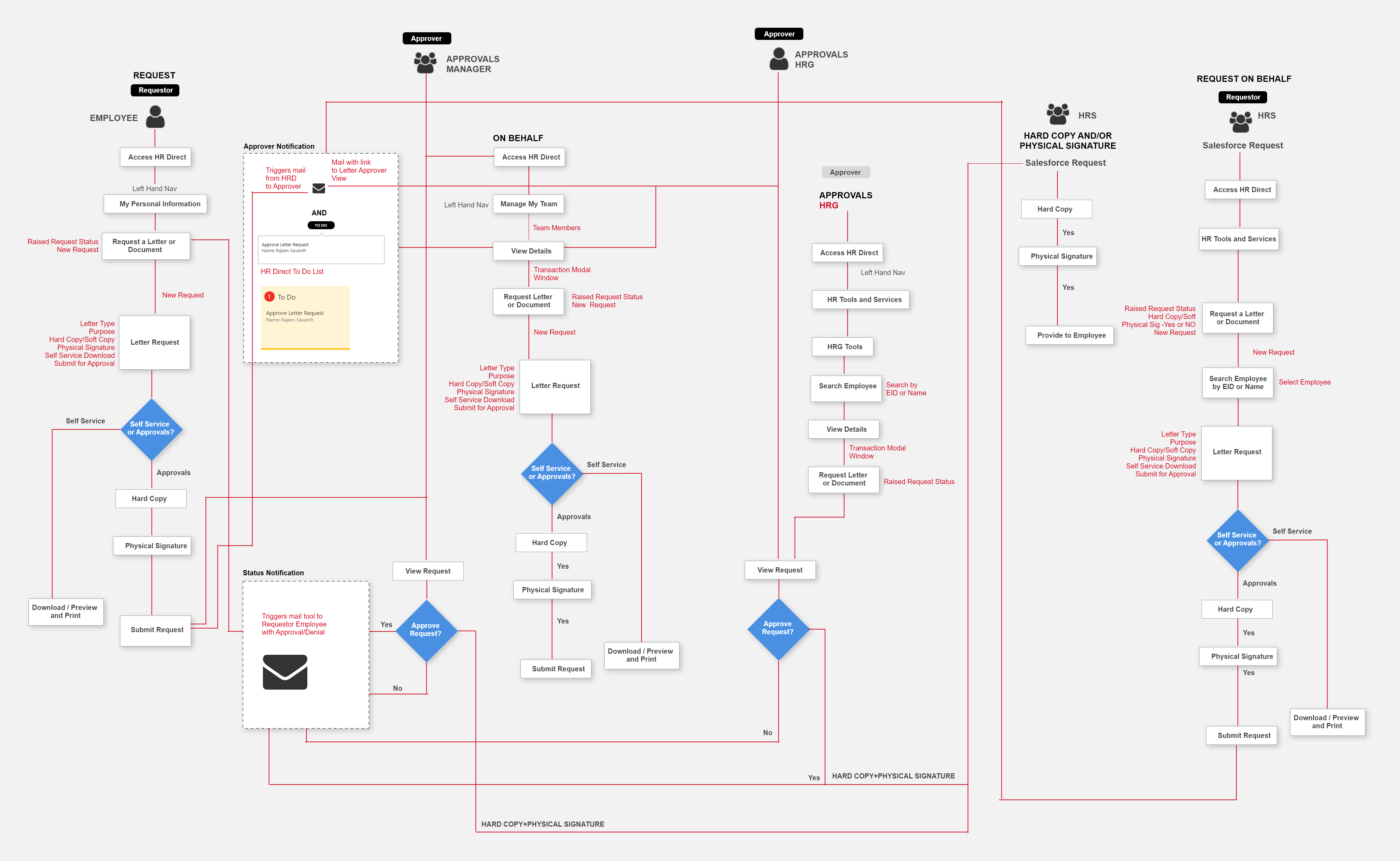
Screen Flow